Genetic Modification
Genetic modification is the modification of genetic material of an organism (DNA/RNA) using a method that does not occur in nature. This page provides guidance on how to safely plan this work at UCL.
- Definitions
Genetic modification is the modification of genetic material of an organism (either DNA or RNA) using a method that does not occur in nature and where the modification can be replicated and/or transferred to other cells or organisms.
Contained use is any activity in which GMOs are cultured, stored, transported, destroyed, disposed of or used in any other way and for which physical, chemical or biological barriers are used to limit contact with humans or the environment to ensure a high degree of safety.
Activity class 1 - Activities of no or negligible risk, for which Containment Level 1 is appropriate to protect human health and the environment
Activity class 2 - Activities of low risk, for which Containment Level 2 is appropriate to protect human health and the environment
Activity class 3 - Activities of moderate risk for which Containment Level 3 is appropriate to protect human health and the environment
Activity class 4 - Activities of high risk for which Containment Level 4 is appropriate to protect human health and the environment
- Before you start work
- You can't start work until your assessment has been reviewed and all appropriate authorisations obtained (including any external approvals from HSE) - you will receive an email notification from riskNET indicating when the assessment has been approved.
- Check that the area where your work is to be carried out is already notified to HSE. If not, contact the University Biological Safety Advisor so that the HSE can be notified. Remember to consider both laboratories and work carried out in Biological Services units when thinking about locations.
- Think carefully about the scope of your assessment. If the activity being assessed is part of a larger project, or connected programme of work, can you assess the whole project / programme rather than generate multiple assessments covering individual activities?
- Remember, your assessment will be seen by others who may not be as familiar with the subject as you, so sufficient detail is required for all those reviewing the assessments (which could include HSE) to understand the nature of the hazards described.
- All statements made about the risks of the work must be justified e.g. with supporting evidence from the Scientific Advisory Committee on Genetic Modification (SACGM) guidance, previous experience or the literature. One word answers are not sufficient. Consider all stages of the planned modification, for example from growing plasmids in a bacterial system, using the plasmids to transfect a cell line, which may or may not also require the use of a viral vector.
If you are not sure which assessment forms to use or need help with completing your risk assessment, speak to your local Genetic Modification Safety Officer in the first instance. You can also contact the University Biological Safety Advisor for further help and advice if needed.
- Using RiskNet
The riskNET tool provides a single means of recording, authorising, filing and archiving your GM risk assessments.
Key features of the system: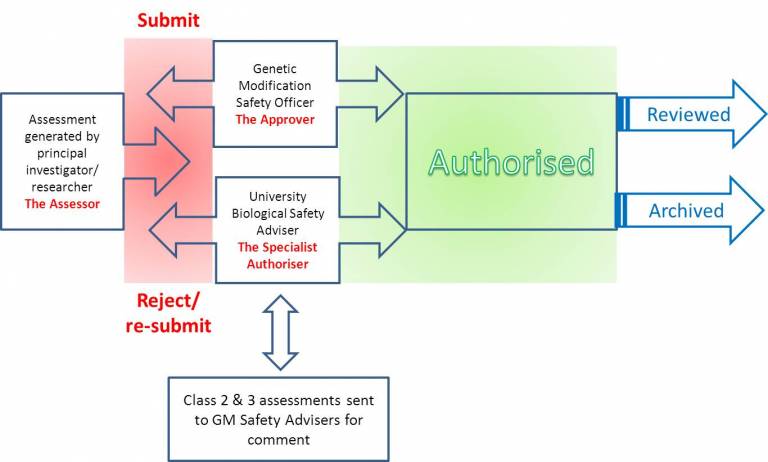
Submission of assessment - automatic generation of unique identification number and automatic email notification of assessment for review is sent to Approver and Specialist Authoriser
Consideration of assessment - Comments recorded on assessment form but also sent in an email notification to assessor(s). The assessor can record their responses on the form as part of the re-submission processes
"Hover-over" technical guidance is available in the riskNET template to help with the completion of assessments. Look for the 'i' icon in a blue circle. Full versions of the guidance are also available in PDF form, for GM animal assessments (pdf) and GM Class 1, 2 and 3 assessments.
After authorisation, email is automatically sent to all those working on a project with a pdf copy of the assessment
Step-By-Step Guides
> Aide memoir for completion of a GM risk assessment pdf
> Drafting and Submitting a GM Risk Assessment pdf
> Reviewing and Approving a GM Risk Assessment (GMSO only) pdf
> Reviewing and Updating a GM Risk Assessment pdf
> Migration of a paper based GM risk assessment to a riskNET specialist pro-forma pdf- HSE notifications (Class 2 and 3 activities only)
In the case of work that requires prior notification to the Health and Safety Executive (HSE) (Class 2 and above), you will need to:
- Complete both the UCL assessment on riskNET and the relevant HSE form (CU2) - use the word form using the following link as it already contains relevant administrative information.
> HSE form (CU2) - Pay a fee to HSE - you will be contacted with a request for your PTA (Project.Task.Award) codes once you have submitted all the relevant paperwork.
> Current HSE fees - All notifications to HSE are made via the University Biological Safety Adviser (USBA). Send your CU2 by email to Safety Services - this will then be filed with your assessment on riskNET once we have received either the acknowledgement or consent from HSE.
When can you start work?
Class 2 - All new and subsequent Class 2 activities must be notified to HSE. As UCL already has Class 2 activities notified to HSE, work may start on receipt of the acknowledgement from HSE unless they indicate that work should not start. You should receive an acknowledgement within 10 (working) days of receipt of the notification by HSE. This will be filed as an attachment with your assessment in RiskNET
Class 3 - Work may not start without written consent from HSE. This will be granted (or refused) within 90 days of the date of the acknowledgement for the first notification of this activity class, and 45 days for subsequent activities.
Work with transgenic plants or animals - Only those activities where the GMO is more harmful than the parental organism need to be notified to HSE. Work can start 45 days after the date of the acknowledgement or earlier if HSE gives written approval for the work to start.
- Complete both the UCL assessment on riskNET and the relevant HSE form (CU2) - use the word form using the following link as it already contains relevant administrative information.
- Containment and control measures for GMM
The containment and control measures needed for work with GMMs drive the Activity classification of your work, so if you only need Containment Level 1 measures to control the risk, then your work is classified as Activity Class 1 and so on.
- Remember you only need to use one measure from the next Level to push the activity up to the next Class.
- You need to use, as a minimum, all Containment Level 1 measures for Class 1 activities (and so on for Class 2-4). However, for notified activities (ie Class 2 and above) you can ask for specific permission not to apply all the measures at a given level. Make sure you that request any reduction in containment at the time of notification to HSE as any subsequent amendments are treated as a significant change to the assessment which will require an additional fee.
- In addition to the specific containment measures needed at any particular level, all work with GMMs should apply the principles of good microbiological practice (GMP) and good occupational safety and hygiene (GOSH).
> The Principles of GMP and GOSH pdf
NB: The containment and control measures necessary for work with GMMs are similar to those needed for work with unmodfied micro-organisms but there are some key differences. If you plan to work with both modified and unmodified micro-organisms, you need to ensure your laboratory meets all necessary requirements - if you need advice on whether your laboratory is suitable for your planned work email: safety@ucl.ac.uk
The following link provides details of containment level 1-4 measures:
- Containment and control measures when for GMO
There are no specified containment levels for work with GM animals or plants as there are for GMMs, but instead the containment and control measures used need to reflect the risks to human health and to the environment if the animal or plant were to escape (e.g. could the animal survive and persist in the environment).
The following link provides a summary of containment measures for work with GM animals and GM plants (from the Scientific Advisory Committee on Genetic Modification - SACGM):
- Techniques not considered to result in genetic modification
Certain techniques are not considered to result in genetic modification:
- in vitro fertilisation;
- natural transformation processes eg conjugation, transduction or transformation; and
- polyploidy induction.
In addition, there are certain techniques where the bulk of the Regulations do not apply, provided they do not use recombinant genetic material or GMOs in the first instance:
- mutagenesis;
- cell fusion (including protoplast fusion) of cells of any eukaryotic species, including the production of hybridomas and plant cell fusions; and
- self-cloning where the resultant organism is unlikely to cause disease or harm to humans.
- Genetic modification safety committee (GMSC)
The UCL Genetic Modification Safety Committee (GMSC) provides advice on all matters relating to genetic modification work and work with hazard group 3 agents at UCL.
It reports to the UCL Health and Safety Committee and is chaired by Professor John Ward, Professor of Synthetic Biology for Bioprocessing in the Department of Biochemical Engineering.
The committee has representatives from Faculties across UCL; including departmental GMSOs, researchers with experience of managing GM work and staff who work at containment level 3.
The committee meets 4 times a year and is principally responsible for ratification of projects previously approved by departments and the University Biological Safety Adviser. It is also responsible for scrutiny and approval of new GM work at class 2 or higher and of new hazard group 3 work.
> Download GMSC's terms of reference including the membership (pdf)
Last updated: Monday, August 3, 2020
 Close
Close


