
Transgenic mice in the study of head induction and development (HS lecture 2)
Gastrulation in the mouse takes place shortly after implantation and is essentially
a similar process to gastrulation in other vertebrates such as Xenopus.
Although the topography is very different the same genetic and cell signalling
pathways appear to be involved.
Mouse development from implantation to mid-gastrulation
Implantation takes place between E4.5 and E6.

Visceral endoderm cells (future amnion, yellow) migrate so that they come to line the entire blastocoel. Trophectoderm (grey) cells adjacent to the ICM proliferate to form the ectoplacental cone and extraembryonic ectoderm. By E6 the epiblast (future embryo, blue) still forms a single layer but has become cup shaped. The whole structure is now termed the egg cylinder.
Gastrulation begins between E6 and E6.5 with the formation of the primitive
streak at the posrterior pole of the epiblast. Epiblast cells that move throught
the streak give rise to mesoderm as in Xenopus. Between E6.5 and E7.5
the streak elongates to the tip of the cylinder and by E8.5 organogenesis has
begun with anterior structures such as heart and brain being the first to develop.
At the anterior end of the streak is a specialised structure called the node,
which is equivalent to the frog dorsal blastopore lip or organiser. Transplantation
of the node has the same effect as that of the organiser (induction of a secondary
axis) and both node and organiser express similar sets of transcription factors
and signalling molecules.
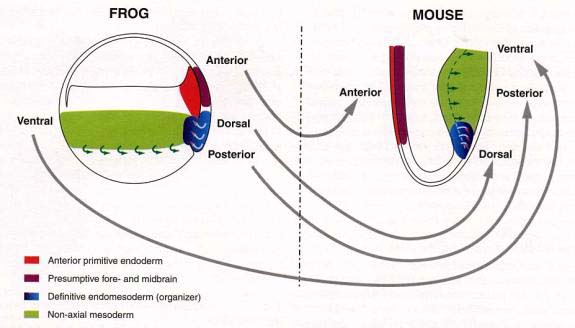
Around mid-gastrulation the mouse embryo can be imagined as as being a Xenopus embryo which has been split open just anterior to the dorsal blastopore lip and turned inside out (see diagram).
Gene function in mouse and Xenopus development
I. Brachyury function in mesoderm development
Methods for studying development in mouse are complemented by those available in Xenopus. In the mouse genes identified by expression or homology
(see above) can be studied by analysing the phenotypic effects of targeted mutations
(loss of function). In some cases genes may also be identified (as in Drosophila)
by the phenotype of mutant animals. Mutations can be spontaneous (e.g. Brachyury-T)
or insertional (e.g. in Nodal).
In Xenopus genes are usually identified by having particular expression
patterns or homology to developmental regulators in other systems. Their function
can then be studied by analysing the effects of overexpression (gain of function),
by injecting mRNA etc.
The Xenopus gene Xbra is expressed transiently in presumptive
mesoderm and later in the notochord. It is one of the first genes to be switched
on when mesoderm inducing factors are added to presumptive ectoderm and injection
of Xbra mRNA itself causes presumptive ectoderm to differentiate as
mesoderm. This suggests but does not prove that Xbra is normally required for
or directs mesoderm development. The genes Brachyury and Tbx6
are mouse homologues of Xbra. Like Xbra, Brachyury
, is expressed in presumptive mesoderm as it passes through the streak and in
notochord. Tbx6 is expressed in mesoderm slightly later than Brachyury
and is absent from notochord. Spontaneous mutations in Brachyury cause
tail defects in heterozygotes. Homozygotes lack notochord and trunk mesoderm.
This confirms that Brachyury is required for mesoderm formation but,
due to the lack of the notochord, the mutants are too disorganised to tell what
has happened to the cells that would have made the trunk mesoderm - do they
just fail to differentiate or do they adopt an alternative fate? Knockouts of
Tbx6 have been made using ES cells. In homozygous mutant for Tbx6
mesoderm is replaced by ectopic neural tubes supporting the later alternative.
II. Organising the anterior-posterior pattern
Organiser/node grafts can induce the formation of a secondary axis in Xenopus
and mice suggesting that signals from the organiser confer anterior-posterior
pattern on both mesoderm and ectoderm. Although early grafts in Xenopus
induce the formation of a complete secondary axis including the head later grafts
only induce trunk structures. This has been interpreted as meaning that separate
signalling pathways organise the head and the trunk. Overexpression of genes
expressed in the organiser to see if this can induce trunk or head formation
have been used to try to identify these pathways. Noggin and chordin
(BMP inhibitors) induce trunk duplications, while nodal related genes induce
complete axes and another gene cerebus induces only head formation.
Cerebus is in fact not expressed in the node proper but just anterior
to it. The equivalent region of the mouse embryo is the anterior visceral endoderm
(AVE) and in the mouse is physically separated from the node. Could contact
with this region induce head identity before cells pass through the node?
A number of genes (nodal, otx2, hex1 and GATA4) are expressed in AVE
before the node forms but later come to be expressed in the node. Knockouts
of otx2, hex1 and GATA4 and an insertional mutant in Nodal
all have very severe effects on head and mesoderm development but this could
be due to their expression in the node. However because the AVE is extraembryonic
it is possible to separate the AVE and node specific effects of these genes
by exploiting the fact that injected ES cells will only contribute to embryonic
tissue. This means that if :-
1) Mutant ES cells are injected into WT blastocysts the AVE will be wild type
and the embryo at least partly mutant.
2) Wild type ES cells are injected into mutant blastocysts the AVE will be completely
mutant and the embryo at least partly wild type.
This type of experiment has been done for Nodal. The results show
that head formation can occur normally as long as the AVE is wild type confirming
that the AVE and not just the node is important for antero-posterior patterning.
References
Gilbert 8th ed. Chapter 11 pp 358-361
Anterior Patterning in Mouse
Beddington R and Robertson E (1998)
Trends in Genetics vol 14 pp 277-284